

Asphyxiation is an invisible hazard, and is the most common cause of fatalities from working with cryogenic liquids. Inert gases have no color, no odor, and no warning properties to alert you of the danger. The only cryogenic liquid that does not pose an asphyxiation hazard is liquid oxygen, for obvious reasons. Liquid nitrogen expands nearly 700 times in volume when it vaporizes. A typical 4 L transfer Dewar of liquid nitrogen, if spilled, can completely displace nearly 2,800 L (~100 cubic feet) of air.
Although the tolerance of individuals for exposure to oxygen deficient environments does vary, the following table outlines some potential effects of acute exposure to various levels of oxygen deficiency:
| Oxygen Concentration | Oxygen Partial Pressure at Sea Level | Potential Effects of Acute Exposure |
|---|---|---|
| 20.9% | 21.2 kPa | Normal atmospheric oxygen concentration |
| ≥ 19.5% | ≥ 19.8 kPa | No adverse effects. Atmosphere is not considered oxygen deficient. |
| < 19.5% | < 19.8 kPa | Mild effects similar to increased altitude. |
| < 17% | < 17 kPa | Reduced night vision, increased breathing volume, accelerated heartbeat, fatigue upon exertion. |
| < 16% | < 16 kPa | Dizziness, increased reaction times, fatigue upon mild exertion. |
| < 15% | < 15 kPa | Impaired attention, impaired judgement, impaired coordination, intermittent breathing, rapid fatigue, loss of muscle control. |
| < 12% | < 12 kPa | Very faulty judgement, very poor muscle coordination, loss of consciousness, permanent heart or brain damage possible. |
| < 10% | < 10 kPa | Inability to move, nausea, vomiting, unconsciousness. |
| < 6% | < 6 kPa | Spasmodic breathing, convulsive movements, death in 5-8 minutes. |
It is also important to note that these effects are based on oxygen concentrations at sea level (1 atm or 101.325 kPa of pressure), but it is the partial pressure of oxygen reaching the lungs that is important, not the volumetric or molar concentration. Therefore at higher altitudes, where the overall air pressure may only be 85 kPa for example, 20.9% oxygen by volume only equates to 17.8 kPa of partial pressure, and individuals who are less tolerant of decreased oxygen pressure may begin to experience adverse effects. The main campus of Lawrence Berkeley National Laboratory sits at about 850 feet above sea level, with a total air pressure of about 98 kPa, and normal oxygen partial pressure of about 20.5 kPa.
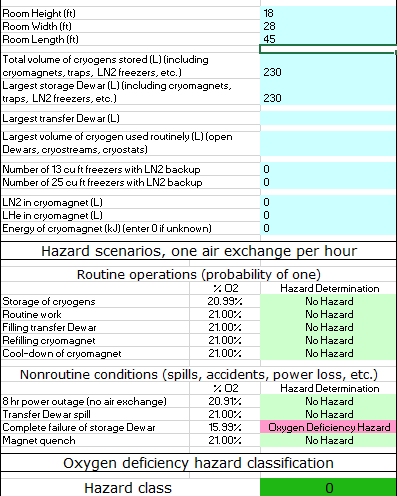
Oxygen deficiency hazard calculations are performed to assess the potential for serious oxygen deficiency in a room where cryogenic liquids are stored and/or handled. Additionally, local areas of severely oxygen deficient atmosphere are possible near open containers and streams of cryogenic liquids. This is especially true with argon, which is a dense gas that tends to accumulate at the floor – even in a room where the oxygen concentration at breathing height is above 20%, the oxygen concentration at the floor may be less than 15%, causing rapid muscle fatigue and potential loss of consciousness.
See the page on Oxygen Deficiency Hazard Analysis for more information about how we mitigate potential asphyxiation hazards.
Download the Oxygen Deficiency Hazard Calculator Tool here, or click on the image to the left.

